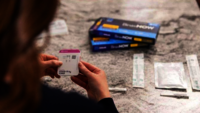
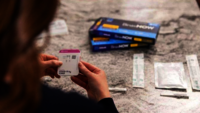
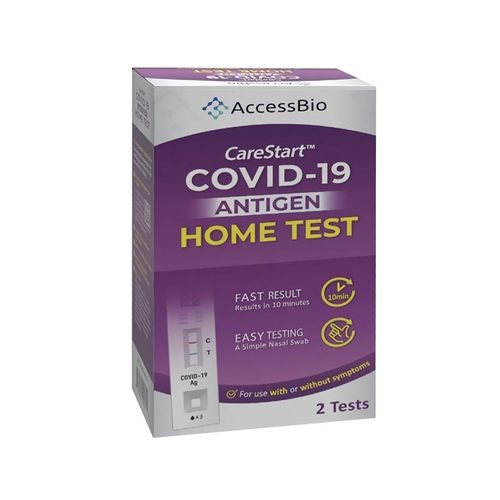
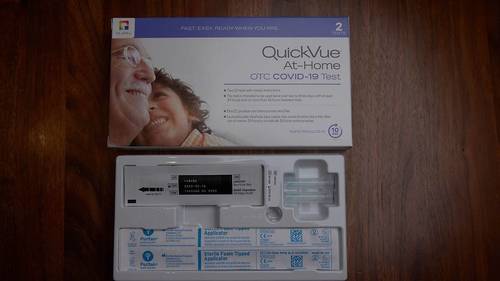
Abbott BinaxNOW At Home Antigen Self Test in Afghanistan
Product Details:
Abbott BinaxNOW At Home Antigen Self Test in Afghanistan Price And Quantity
- 1 Box
- 1.20 - 1.50 USD ($)/Box
- 1.2-1.5 USD ($)/Box
Abbott BinaxNOW At Home Antigen Self Test in Afghanistan Trade Information
- USA
- 3 Box Per Day
- 5-15 Days
- South America Middle East Central America Eastern Europe Asia Western Europe North America Australia Africa
- Abbott BinaxNOW At Home Antigen Self Test in Afghanistan.FDA EUA CE ISO GMP SFC ETC
Product Description
BinaxNOW COVID-19 Antigen Self-Test Home Kit is a simple solution for COVID-19 infection detection,Abbott BinaxNOW At Home Antigen Self Test in Afghanistan with rapid results in the convenience of your home. This test has received FDA Emergency Use Authorization for self-testing without the need to ship samples to a lab or for a prescription from your healthcare provider. This 15-minute test can be completed anytime, anywhere. Simply test yourself twice within 3 days, with at least 36 hours between tests. Each Kit contains 2 tests
The BinaxNOW COVID-19 Antigen Self Test has not been FDA cleared or approved. It has been authorized by the FDA under an emergency use authorization. It has been authorized only for the detection of proteins from SARSCoV-2, not for any other viruses or pathogens, and are only authorized for the duration of the declaration that circumstances exist justifying the authorization of emergency use of in vitro diagnostics for detection and/or diagnosis of COVID-19 under Section 564(b)(1) of the Federal Food, Drug and Cosmetic Act, 21 U.S.C.Abbott BinaxNOW At Home Antigen Self Test in Afghanistan 360bbb-3(b)(1), unless the declaration is terminated or authorization is revoked sooner. BinaxNOW COVID-19 Antigen Self Test should be performed twice in 3 days, at least 36 hours apart
 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese