


Quickvue at-home otc covid-19 test kit in Iran
Product Details:
Quickvue at-home otc covid-19 test kit in Iran Price And Quantity
- 1.20 - 1.50 USD ($)/Bag
- 1 Box
- 1.2-1.5 USD ($)/Bag
Quickvue at-home otc covid-19 test kit in Iran Trade Information
- USA
- 3 Box Per Day
- 5-15 Days
- Western Europe Australia North America Eastern Europe Middle East Central America South America Asia Africa
- Quickvue at-home otc covid-19 test kit in Iran.FDA EUA CE ISO GMP SFC ETC
Product Description
Why choose Quidel QuickVue At-Home OTC COVID-19 Test Kit?
The test is intended to be used twice over two to three days, with at least 24 hours and no more than 36 hours between tests
Positive results indicate the presence of viral antigens, Quickvue at-home otc covid-19 test kit in Iran but clinical correlation with past medical history and other diagnostic information is necessary to determine infection status
Negative results should be treated as presumptive, do not rule out SARS-CoV-2 infection and should not be used as the sole basis for treatment or patient management decisions, including infection control decisions
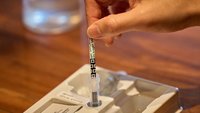
The test is intended to be read at 10 minutes; If the test is read before this or is read more than 5 minutes after the indicated read time, results may be inaccurate and the test should be repeated.
For In Vitro Diagnostic (IVD) Use
This kit has not been FDA cleared or approved but has been authorized by FDA under an EUA.
It has been authorized only for the detection of proteins from SARS-CoV-2, not for any other viruses or pathogens.
The emergency use of this covid 19 test kit is only authorized for the duration of the declaration that circumstances exist justifying the authorization of emergency use of IVDs for detection and/or diagnosis of COVID-19 under Section 564(b)(1) of the Federal Food, Drug and Cosmetic Act, 21 U.S.C. 360bbb-3(b)(1), unless the declaration is terminated or authorization is revoked sooner.
Quickvue at-home otc covid-19 test kit in Iran
QuickVue Test Kit Safety Information
This test kit is intended for the qualitative detection of the nucleocapsid protein antigen from SARS-CoV-2 from individuals with or without symptoms of COVID-19. The test is intended to be used twice over two to three days,with at least 24 hours and no more than 36 hours between tests.
Read the written instructions fully before starting the test procedure
Quickvue at-home otc covid-19 test kit in Iran
To ensure correct results, you must follow the instructions
Keep test kit and materials out of the reach of children and pets before and after use
Wear safety mask or other face covering when collecting swabs from children or others
Use of personal protection materials such as gloves are recommended
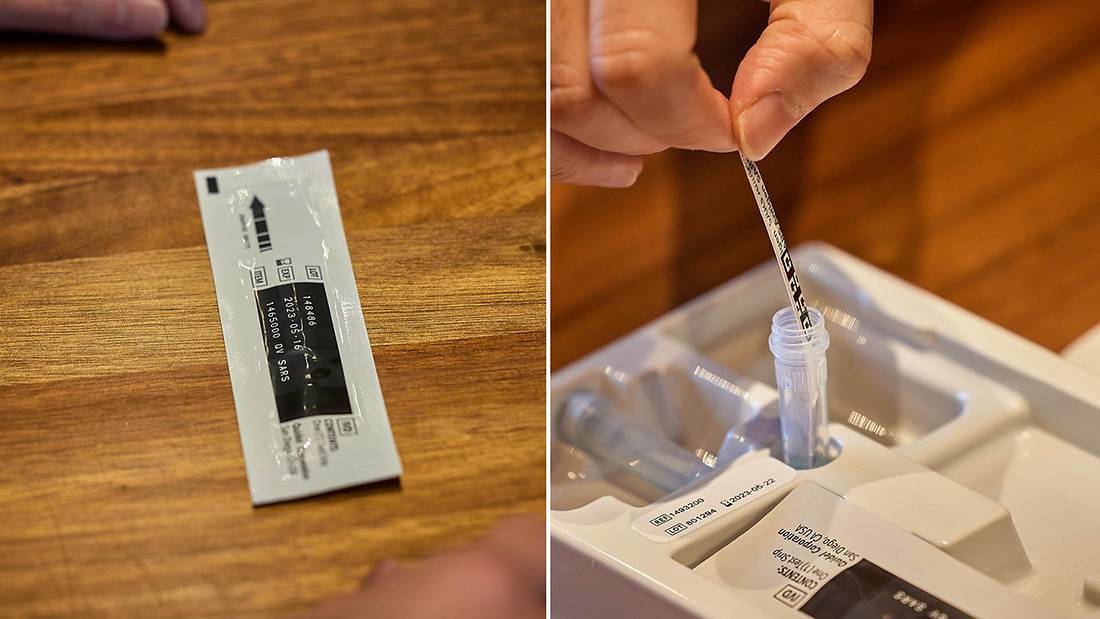
Do not open the materials until ready for use. If the test strip is open for an hour or longer, invalid test results may occur.
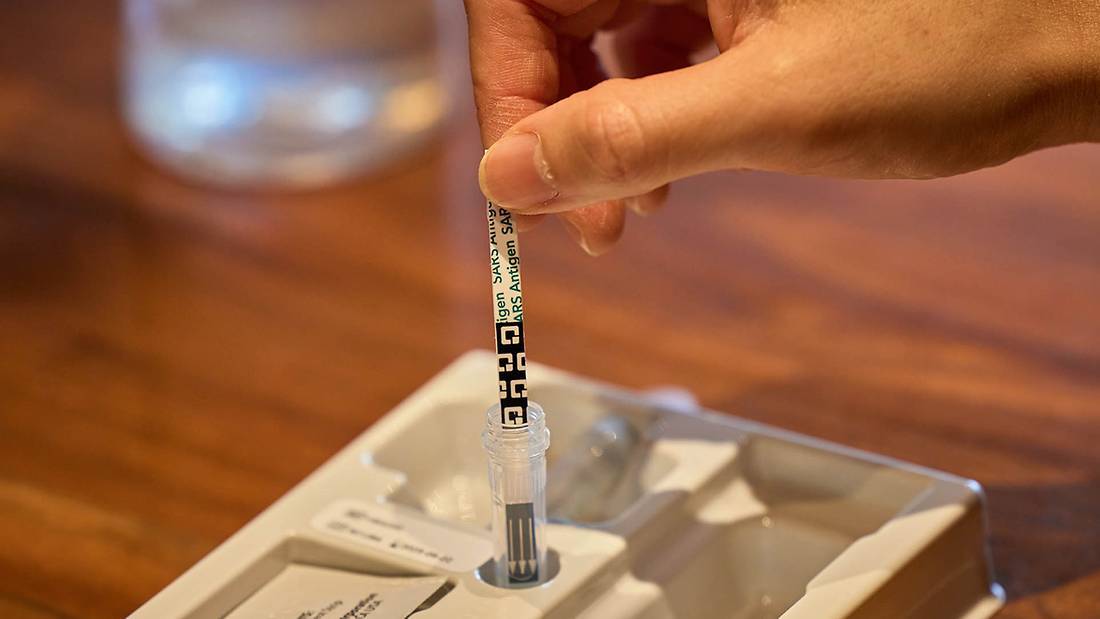
Improper swab collection may result in incorrectly negative (false negative) results
The test is intended to be read at 10 minutes. If the test is read before this or is read more than 5 minutes after the indicated read time, results may be inaccurate and the test should be repeated.
Do not use a test kit that is expired
Do not touch the swab head when handling the swab
Avoid exposure of your skin, eyes, nose, or mouth to the solution in the tube.
 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese